
Mental Health for Pompe Disease: The Importance of Being Understood
Title: Mental Health for Pompe DiseaseA rare genetic disease in which the body cannot properly break down glycogen, leading to buildup tha... More: The Importance of Being Understood
Date: Thursday, May 14, 2026
Time: 12 p.m. CT / 1 p.m. ET
Speakers:
Heather Shorten
Nadia Bodkin, Pharm. D., MS
JC Muyl
Summary:
Living with Pompe diseaseA rare genetic disease in which the body cannot properly break down glycogen, leading to buildup tha... More means navigating far more than the physical realities of a complex, progressive condition. The emotional and psychological toll, including the uncertainty, the isolation, and the challenge of finding care that truly sees the whole person, is a dimension of this journey that is too often left unaddressed. This webinar brings together three advocates at the forefront of mental health and rare disease: Heather Shorten of Pompe Alliance, Nadia Bodkin of the Rare Advocacy Movement, and JC Muyl of Mental Health For Rare.
Drawing on community research and lived experience, our presenters will explore the unique emotional challenges tied to living with Pompe diseaseA rare genetic disease in which the body cannot properly break down glycogen, leading to buildup tha... More, why feeling understood matters so deeply, and what meaningful mental health support can look like for this community. Whether you are a patient, caregiver, healthcare provider, or advocate, this is an opportunity to be part of an important and long-overdue conversation about whole-person care in the Pompe diseaseA rare genetic disease in which the body cannot properly break down glycogen, leading to buildup tha... More community.

Move How You Can, Rest All You Need
Hey, all! Lucas Garrett, here. As we head into April, I wanted to take a minute to jot down a few ideas on staying healthy — physically and mentally. As many in the Pompe community may already be keenly aware, well-regulated habits of getting quality sleep, eating...

Immune Responses to AAV Gene Therapy
Title: Immune ResponsesThe body's natural defense system reacting to substances it sees as foreign, including treatments li... More to AAVA harmless virus commonly used as a delivery system in gene therapy to carry healthy genes into cell... More Gene TherapyA developing treatment that aims to fix, replace, or add genetic instructions. More: Challenges and Emerging Solutions
Date: Thursday, April 23, 2026
Time: 12 p.m. CST / 1 p.m. EST
Speaker:
Abigail Benkert, MD
Summary:
Immune responsesThe body's natural defense system reacting to substances it sees as foreign, including treatments li... More to AAVA harmless virus commonly used as a delivery system in gene therapy to carry healthy genes into cell... More gene therapyA developing treatment that aims to fix, replace, or add genetic instructions. More remain a major barrier to its access and effectiveness. This talk will highlight key immune challenges and emerging strategies to overcome them, including antibody-cleaving enzymes and novel capsids designed to evade pre-existing immunity.
Honoring Advocacy: Celebrating Tiffany House and the RareVoice Awards
This week is Rare Disease Week, with events and activities taking place worldwide to bring awareness to rare diseases and honor the community. One such event is "Rare Disease Week on Capitol Hill," where Tiffany House is being featured as a RareVoice Awardee for State...

Tiffany House Receives RareVoice Award for State Advocacy
We are honored to share this moment recognizing Tiffany House as a recipient of the RareVoice Award for State Advocacy. The award is presented by the EveryLife Foundation For Rare Diseases.This award is given to advocates whose voices have helped shape meaningful...

2026 PCMA Pull for Pompe Fundraiser
The AMDA is excited to announce that the 15th Annual PCMA's Pull for Pompe fundraiser will take place on Saturday, May 2, 2026, from 8 a.m. to 2 p.m. CST at the National Shooting Complex in San Antonio, Texas! The Pull for Pompe clay shooting event is designed for all...

Making Sense of Research Studies and Registries
Title: Making sense of research studies and registries
Date: Thursday, February 19, 2026
Time: 7 PM CST / 8 PM EST
Speakers:
Karmen Trzupek
Ryan Colburn
Summary:
Have you heard about research studies and patient registries for Pompe diseaseA rare genetic disease in which the body cannot properly break down glycogen, leading to buildup tha... More? These studies are a critical tool in capturing the patient experience, which improves disease understanding, management, and treatment options.
Join this session to learn about the different types of research studies and patient registries for Pompe diseaseA rare genetic disease in which the body cannot properly break down glycogen, leading to buildup tha... More. What are the differences between them? How is the data shared and used? We’ll cover what you may want to consider when choosing to participate, cover a few of the historical registries that have contributed to progress in Pompe, and share an innovative patient led registry, Progress4Pompe, where patients own their data and choose how to share it.

With Gratitude: Honoring Our 2025 Donors
The Acid Maltase Deficiency Association (AMDA) is deeply grateful to the individuals, families, and organizations who contributed in 2025. Your generosity makes it possible for us to fund research and promote public awareness of Acid Maltase Deficiency, also known as Pompe diseaseA rare genetic disease in which the body cannot properly break down glycogen, leading to buildup tha... More.

New Year’s Resolutions and Hobbies
Are New Year’s resolutions already starting to feel heavy? Lucas explores why we put so much pressure on ourselves, and why breaking a resolution can spiral into guilt. His gentler reset is simple: pivot to a hobby, and if it does not bring you joy, you can stop without feeling bad about it. He also points to research linking hobby engagement with mental health and well-being.

Grief Happens in Rare Disease: Let’s Talk About it Already
Title: Grief Happens in Rare Disease: Let’s Talk About it Already
Date: Thursday, January 15, 2026
Time: 1 PM CST / 2 PM EST
Speaker:
Lisa Keefauver, MSW
Webinar Overview:
Given 100% of us experience loss, multiple times in our lives, means that when we work with humans, we’re working with grievers. That’s why our grief-illiterate culture is deeply problematic, causing so much unnecessary suffering. Based on her professional wisdom as a narrative-therapy trained social worker and her personal insights as a widow and cancer survivor, Keefauver combines metaphor, science, storytelling and humor to help audiences more fully understand the expansiveness and pervasiveness of both the sources and impact of grief in our work and personal lives.

Burnout, Creativity, and Rest
AMDA’s Lucas Garrett shares a thoughtful reflection on burnout and creativity — exploring how stepping back to rest can reignite inspiration and help restore a sense of balance, both in art and everyday life.

Tiffany House and Maryze Schoneveld van der Linde Honored at Sanofi’s Global Rare Impact Awards
Sanofi’s inaugural Global Rare Impact Awards recognized three pioneers in rare disease advocacy, including the AMDA’s late president Tiffany House and IPA board member Maryze Schoneveld van der Linde.

Morgan’s Story: When Hope Met Hurricane Katrina
Editor's NoteIn the early years of Pompe research, access to enzyme replacement therapy (ERT)A treatment that replaces the missing enzyme through IV infusion. More was extremely limited. Long before FDA approval, a handful of families, physicians, and researchers worked tirelessly to secure treatment for patients diagnosed with Pompe...

Check Back Soon for Upcoming Webinars
We don’t have a webinar scheduled at the moment, but more exciting sessions are on the way! Check back soon for details, or sign up for our newsletter to be notified when new webinars are announced.

Overview of Breathing Muscle Weakness in Neuromuscular Disease (NMD)
Title: Overview of Breathing Muscle WeaknessA loss of strength that can affect movement, posture, and sometimes breathing. More in Neuromuscular Disease (NMD)
Date: Thursday, September 25, 2025
Time: 1 PM CST / 2 PM EST
Speaker:
Andrea Klein
Webinar Overview:
An introduction to the concepts of breathing and coughing muscle weaknessA loss of strength that can affect movement, posture, and sometimes breathing. More, this presentation highlights the importance of medical self-advocacy to ensure positive outcomes. Topics include the role of muscles in breathing, the relevance of blood gases, the evaluation process, appropriate interventions, and more.

Grief and Rare Disease: Recognizing Loss and Building Grief Literacy
This past May, our hearts broke twice. Within just two weeks, the Pompe community said goodbye to two extraordinary advocates: Maryze Schoneveld van der Linde with the IPA, and our beloved Tiffany House with AMDA/IPA. Their passing illuminated something profound: grief lives everywhere in our community, often in ways we don’t recognize or name.
The parent who mourns the carefree childhood their child will never have. The adult with late-onset Pompe diseaseA form of Pompe disease that begins after infancy and usually progresses more slowly. More who grieves the marathon they’ll never run again. The spouse watching their partner’s strength fade, carrying the weight of tomorrow’s uncertainties. These experiences are real, valid, and profound, yet they often go unacknowledged. We call this grief illiteracy, and it’s time we changed that conversation.
Grief isn’t weakness. It’s love with nowhere to go. It’s hope recalibrating. It’s proof that something mattered deeply to us. When we learn to recognize grief’s many forms and respond with compassion, we create space for authentic healing and build a community where no one has to carry their sorrow alone.
Read the full article to explore the different types of grief, understand how it affects our daily lives, and discover practical ways to build grief literacy in the Pompe community.
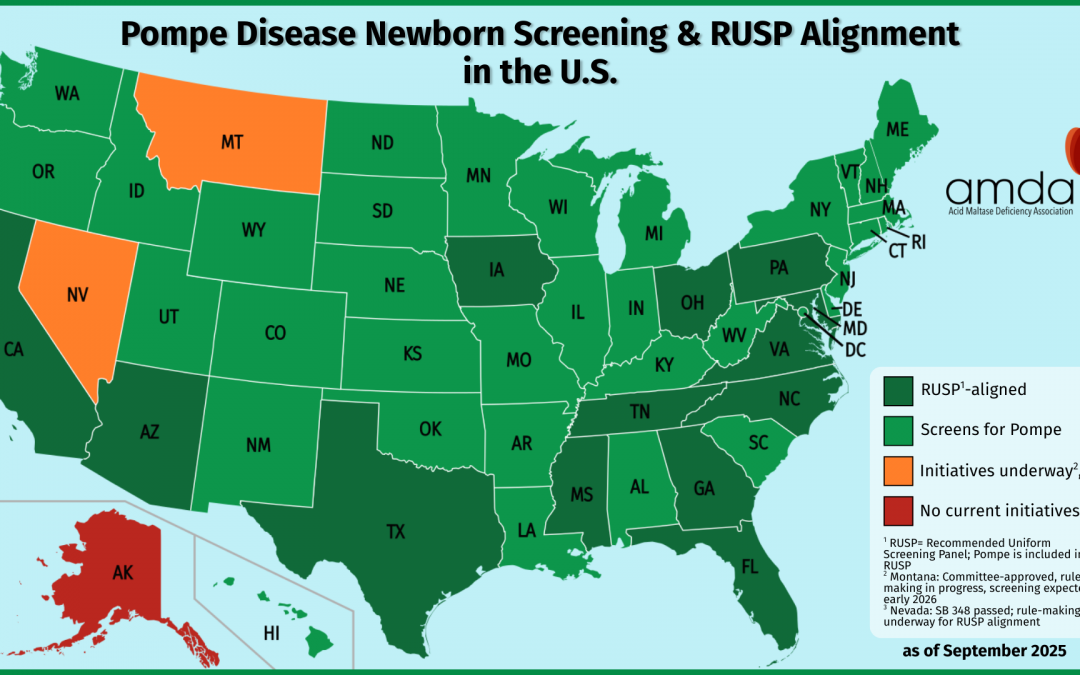
Closing the Gaps: Pompe Disease Newborn Screening and the Importance of RUSP Alignment
A Milestone for Pompe Families This SeptemberSeptember is Newborn Screening Awareness Month, a time to celebrate the programs that ensure every baby has a healthy start. This year, there's especially good news for families affected by Pompe diseaseA rare genetic disease in which the body cannot properly break down glycogen, leading to buildup tha... More: on August 18,...
Three Decades of Impact: Celebrating 30 Years of the AMDA
A Legacy of Leadership, Love, and Lifesaving Work in the Pompe Community This August, the Acid Maltase Deficiency Association (AMDA) turns 30. Thirty years of progress. Thirty years of advocacy. Thirty years of showing up for patients and families around the world....

2024 Helen Walker Research Grant Recipient: Dr. Jaymin Upadhyay, Boston Children’s Hospital, Harvard Medical School
The winner of the 2024 AMDA Helen Walker Research Grant: Dr. Jaymin Upadhyay, Boston Children’s Hospital, Harvard Medical School Project Title: Implementation of At-Home Functional BiomarkerA measurable sign in the body that helps track disease progression or treatment response. More Methods in Patients with Pompe DiseaseA rare genetic disease in which the body cannot properly break down glycogen, leading to buildup tha... More. For the second year in a row, the...

Texas Adds Pompe Disease to Newborn Screening Panel—A Historic Milestone Coinciding with the AMDA’s 30th Anniversary
Great news for the rare disease community—Texas has taken a big step by adding Pompe diseaseA rare genetic disease in which the body cannot properly break down glycogen, leading to buildup tha... More to its newborn screening panel! This major advancement in protecting the health of Texas infants also marks a meaningful moment for the Acid Maltase Deficiency Association...
